Stem Cell Therapy for Anti-Aging: Separating Evidence from Hype
Quick Takeaway
Established Science: Stem cell therapy has clear, FDA-approved uses in blood cancers and certain immune conditions.
The “Reset” Myth: Evidence for whole-body anti-aging or “biologic reset” effects remains early and limited.
International Data: Regulatory hubs in Japan and the Caribbean are generating cumulative real-world safety data for "elective" use, though they lack long-term, placebo-controlled proof.
The Mechanism: Most longevity protocols use mesenchymal stem cells that act as immune “managers,” not replacement parts.
The Cost: Private clinic treatments often range from $5,000 to $25,000 and are typically not covered by insurance.
Bottom Line: The biology is intriguing, but the long-term safety profile for elective anti-aging use is not yet fully defined.
Why Stem Cell Therapy Generated So Much Hope
Few areas of medicine have captured the public imagination like stem cell therapy. The idea is simple and powerful: if aging and chronic disease reflect accumulated cellular damage, then introducing regenerative cells might help restore or stabilize those systems.
In laboratory research, stem cells influence inflammation, immune signaling, and tissue repair. That biological plausibility created a compelling narrative—not just managing disease, but repairing it. High-profile biohackers and longevity influencers have further amplified interest in these therapies, often racing ahead of peer-reviewed consensus.
For patients with cancer, heart disease, autoimmune illness, or degenerative joint disease, that possibility is deeply attractive. In longevity medicine, the question expands further: Could regenerative therapies slow aspects of biological aging itself?
Hope grew quickly. Evidence has grown more cautiously.
What Stem Cells Actually Are
The term “stem cell therapy” is often used broadly, but it refers to different types of cells with very different functions.
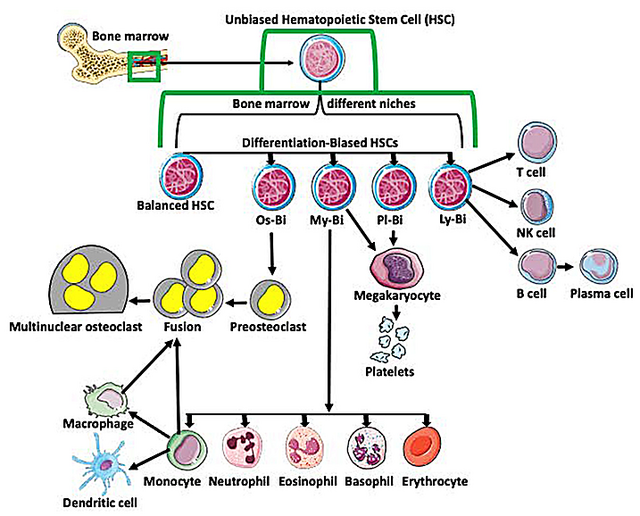
1. Hematopoietic Stem Cells (HSCs)
Hematopoietic stem cells are blood-forming stem cells found in bone marrow. They are the "builders" of the blood system.
They produce:
Red blood cells (oxygen carriers)
White blood cells (immune defenders)
Platelets (clot formers)
When someone receives a bone marrow transplant for leukemia, they are receiving hematopoietic stem cells. This therapy has decades of strong clinical evidence and remains standard of care.¹ This is established medicine—not speculative longevity treatment.
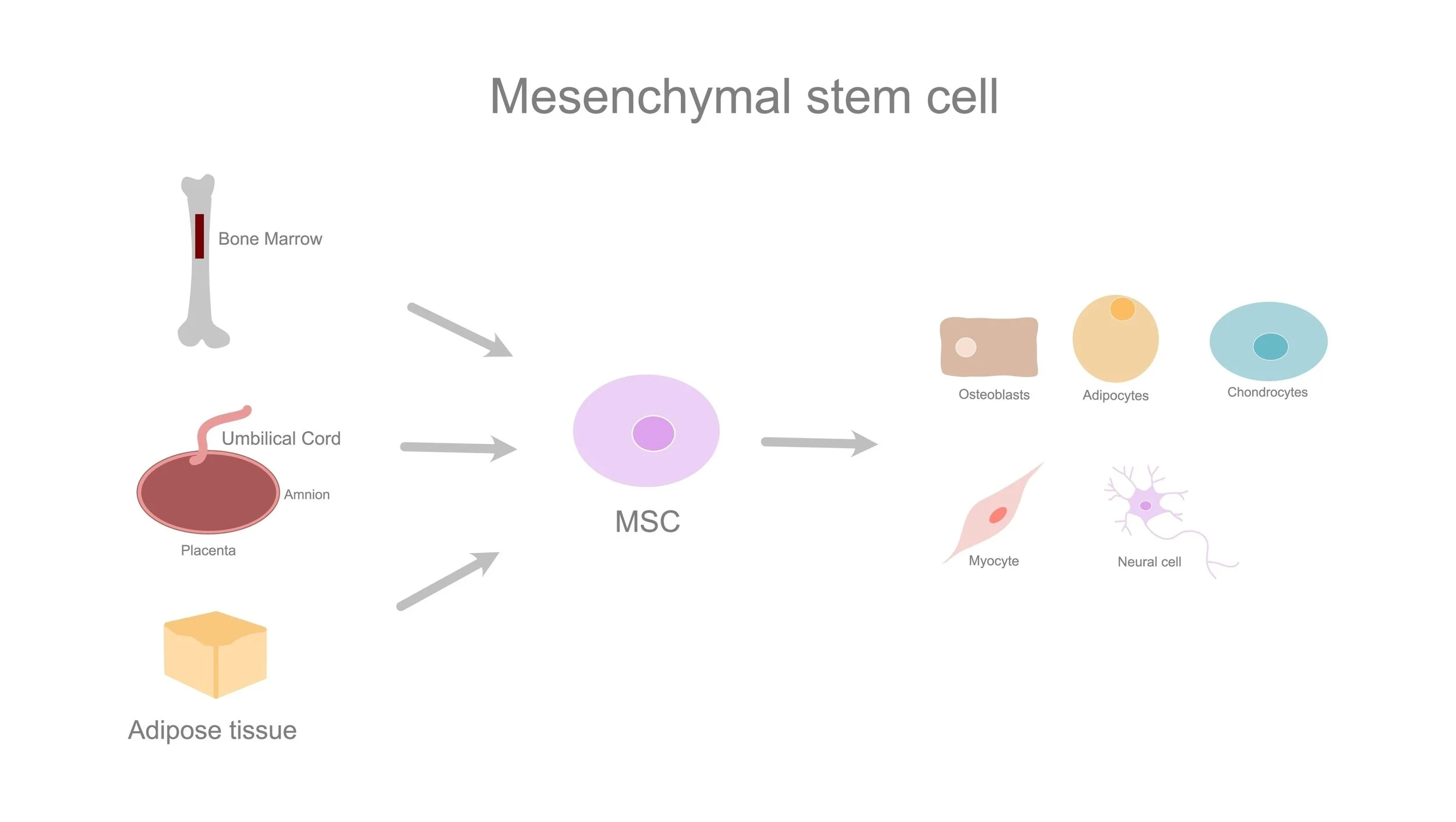
2. Mesenchymal Stem (or Stromal) Cells (MSCs)
Most longevity clinics are not using blood-forming stem cells. They are using mesenchymal stem cells (MSCs).
Mesenchymal stem cells can be derived from:
Bone marrow
Adipose (fat) tissue
Umbilical cord tissue
Unlike hematopoietic stem cells, mesenchymal stem cells do not primarily function by turning into new organs or rebuilding damaged structures. Instead, they act largely through paracrine signaling—meaning they release chemical signals that influence nearby cells.²
A Simple Analogy
Think of mesenchymal stem cells as site managers, not replacement bricks.
They release molecules (including cytokines and exosomes) that tell your existing cells to lower inflammation and start repairing themselves. Most intravenously infused MSCs do not permanently integrate into organs; many are temporarily trapped in the lungs and cleared within days.³ Their benefit lies in immune modulation—not structural regeneration.
[Quote Block or Callout] A Brief Note on Exosomes: Because MSCs work primarily by signaling, some newer therapies attempt to use exosomes alone—tiny vesicles containing those signaling molecules. This is an active research area but currently carries similar regulatory and evidentiary uncertainties as live cell therapy.
Where the Evidence Is Strong
1. Blood Cancers and Bone Marrow Failure Hematopoietic stem cell transplantation remains a life-saving, well-established therapy in leukemia, lymphoma, and bone marrow disorders.¹
2. Graft-versus-Host Disease Graft-versus-host disease occurs after a bone marrow transplant when donor immune cells attack the recipient’s tissues. MSCs have demonstrated benefit in certain cases of steroid-resistant graft-versus-host disease, representing a validated immunomodulatory application.⁴
3. Perianal Crohn’s Fistulas Crohn’s disease is a chronic inflammatory bowel condition. Some patients develop fistulas—abnormal tunnel-like connections between the intestine and surrounding tissue. Randomized trials show that locally injected MSCs can improve healing in this specific condition.⁵
Key Research Findings: What Trials Actually Suggest
Recent clinical trials offer a clearer picture of what mesenchymal stem cell therapy may—and may not—accomplish.
Brain Health & Alzheimer’s Disease (The CLEAR MIND Trial)
In company-reported Phase 2a data presented in 2024, patients with mild Alzheimer’s disease received an allogeneic (donor) MSC product.¹¹
The Findings: The therapy met safety endpoints. More interestingly, MRI imaging suggested slower shrinkage (atrophy) of the hippocampus compared to placebo.
The Limitations: This was a small, early-phase trial not powered to prove cognitive reversal. It represents a biological "signal," not proof of a cure.
Chronic Heart Failure (The DREAM-HF Trial)
The DREAM-HF Phase 3 trial enrolled over 500 patients with chronic heart failure.¹²
What It Didn't Do: The therapy did not significantly reduce recurrent hospitalizations. It did not structurally repair the weakened heart muscle enough to restore pumping function (ejection fraction, the percentage of blood pumped out with each heartbeat).
What It Did Do: Secondary analyses suggested a reduction in major adverse cardiovascular events in certain subgroups, though these findings were not primary endpoints and require cautious interpretation.
Interpretation: This reinforces the immune-modulation “manager” model. The cells likely reduced systemic inflammation (preventing strokes) even though they failed to "regrow" the heart.
Aging Frailty (The CRATUS Projects)
Frailty refers to a measurable syndrome of reduced strength, endurance, and physiologic reserve in older adults.
The Findings: Earlier Phase 1/2 trials (2017) demonstrated that MSC infusions were safe and improved 6-minute walk distance performance in frail older adults.⁹
The Context: While promising, these benefits were often temporary, leading many current protocols to explore repeat dosing.
Knee Osteoarthritis (2024 Meta-Analysis)
A 2024 systematic review examined MSC injections for knee osteoarthritis.¹³
The Findings: Significant pain reduction and improved function for up to 12 months.
The Limitations: Benefits often diminished by 24 months, with no convincing evidence of cartilage regrowth. This suggests a temporary biological "boost" rather than a permanent fix.
What About Data from International Regenerative Hubs?
Patients frequently notice that stem cell clinics are more common in countries such as Japan, Panama, the Cayman Islands, Switzerland, and parts of the Caribbean. The assumption is often one of two extremes: either these locations are “more advanced,” or they are operating in a regulatory vacuum.
The reality is more nuanced. Different countries have adopted different regulatory philosophies around regenerative medicine.
Japan: The Conditional Approval Model
Japan implemented the Act on the Safety of Regenerative Medicine in 2014. Rather than requiring large Phase 3 trials before commercialization (as the United States typically does), Japan allows conditional approval for certain regenerative therapies if early studies demonstrate safety and preliminary evidence of benefit.
What This Has Produced: Substantial real-world safety data for autologous (patient-derived) adipose stem cell procedures, structured oversight, and data collection.
What It Has Not Produced: Large placebo-controlled longevity trials or definitive evidence of systemic anti-aging effects. The Japanese model accelerates access—but does not eliminate uncertainty.
Panama and Central America: Observational Data
Some international centers in Central America operate under national regulatory frameworks that allow expanded use of mesenchymal stem cells. Several have published Phase I/II safety studies and open-label trials in neurological/autoimmune conditions.
The Findings: These studies frequently demonstrate acceptable short-term safety and reductions in inflammatory markers.
The Caveat: Many are open-label (no placebo group). Selection bias is significant, and lifestyle changes often occur simultaneously. This limits the strength of the conclusions.
Cayman Islands & The Bahamas: High-End Longevity Protocols
Some Caribbean jurisdictions have structured health commissions requiring licensed physicians and accredited facilities. These centers often focus on intravenous MSC infusions and “systemic inflammation” protocols.
The Data: Observational registry data commonly report short-term reductions in inflammatory markers (e.g., CRP) and subjective energy improvements lasting 3–6 months.
The Limitation: Most data are registry-based with no placebo control and no long-term cancer or mortality follow-up. These patterns are consistent with transient (short-term) immune modulation rather than durable systemic reversal.
Switzerland: Biotech Infrastructure
Switzerland has a strong biotech research ecosystem and regulatory pathways that can be somewhat more flexible than the U.S. FDA’s biologics system. This has enabled Advanced Therapy Medicinal Product (ATMP) development and high-standard cell expansion facilities. However, as with Japan, the presence of infrastructure does not equal proof of longevity benefit.
What International Experience Actually Suggests
When viewed soberly, the international data supports three cautious conclusions:
Short-term safety of properly processed mesenchymal stem cell infusions appears acceptable in controlled settings.
Transient anti-inflammatory effects are biologically plausible and observed.
Many protocols recommend repeat infusions at 6–12 month intervals, underscoring that any observed benefit may not be durable.
Safety & Risks: What Critics Are Right About
While the potential of stem cells is exciting, critics and regulatory bodies (like the FDA) raise valid concerns that every patient should understand.
1. The Cancer Question (Tumorigenicity)
A common fear is that stem cells, which are designed to grow, might grow uncontrollably and form tumors.
The Nuance: This risk is primarily associated with embryonic or induced pluripotent stem cells, which can form teratomas (tumors of mixed tissue).
MSC Reality: Mesenchymal stem cells (MSCs) are "multipotent" (limited) and have an excellent safety record regarding tumor formation in human trials.
The "Fertilizer" Risk: However, because MSCs secrete growth factors, there is a theoretical risk they could stimulate the growth of existing pre-cancerous cells. This is why reputable clinics strictly screen patients and often refuse to treat those with active cancer or a recent history of malignancy. Long-term oncologic surveillance data for elective anti-aging use remain limited.
2. Infection & "Dirty Labs"
The most common serious complication in real-world reports is not cancer, but infection. In 2018, the CDC reported that at least 12 patients across three states were hospitalized with severe bacterial infections (E. coli, Enterobacter) after receiving contaminated stem cell products from a non-GMP distributor.¹⁴
Takeaway: This underscores why asking for GMP (Good Manufacturing Practice) certification is non-negotiable.
3. Ectopic Tissue (Growth in the Wrong Place)
If cells are injected into areas where they don't belong, they can sometimes behave unpredictably. There are documented cases, such as a patient developing small bone fragments in her eyelid after receiving a stem cell injection around the eye.
Takeaway: Never let a provider inject bone marrow or fat-derived cells into sensitive areas (like the eyes) without high-level specialist guidance.
Why Large Longevity Trials Are So Difficult
If the science is promising, why don’t we have definitive proof of anti-aging effects?
Time: Humans live for decades. Proving healthspan extension requires very long follow-up.
Complexity: Aging depends on genetics, diet, sleep, exercise, environment, and socioeconomic factors.
Imperfect Proxies: Researchers rely on markers such as inflammatory levels or imaging changes. We do not yet know how reliably these translate into longer lifespan.
The “Pay-to-Participate” Model vs Clinical Trials
A critical distinction for patients:
Regulated Clinical Trials: Participants do not pay. There are placebo groups. Independent review boards oversee safety.
"Registries" & Clinics: Some private clinics label treatments as “studies” or “registries” while charging patients thousands of dollars. While these registries can collect useful data, they are not equivalent to randomized controlled trials.
If You Are Considering Stem Cell Therapy: What to Ask
If you explore this path, ask these technical questions to vet the clinic:
Source (Autologous vs. Allogeneic): Are these your cells (autologous) or donor cells (allogeneic)? Allogeneic means donor-derived; autologous means derived from your own body. Donor cells may be "younger," but your own cells carry zero rejection risk.
Passage Number: How many times were the cells divided in the lab? Lower numbers generally indicate more potent cells.
Viability: What percentage of the cells are actually alive at the moment of infusion? (If viability is 60%, you are getting nearly half the intended dose).
GMP Certification: Is the lab certified for Good Manufacturing Practice? This is non-negotiable for safety and sterility.
Bottom Line
Stem cell therapy is a legitimate and evolving field. Its established uses in cancer and immune disease are life-saving. Its potential in longevity is biologically plausible and supported by early signals.
However, proven systemic anti-aging effects do not yet exist. Be cautious of guarantees, understand the difference between experimental therapy and validated treatment, and approach with informed discernment.
References
Appelbaum FR. Hematopoietic-cell transplantation at 50. N Engl J Med. 2007.
Caplan AI. Mesenchymal stem cells: time to change the name? Stem Cells Transl Med. 2017.
Eggenhofer E, et al. The life and fate of mesenchymal stem cells. Front Immunol. 2014.
Le Blanc K, et al. Mesenchymal stem cells for steroid-resistant acute graft-versus-host disease. Lancet. 2008.
Panés J, et al. Expanded allogeneic adipose-derived mesenchymal stem cells for Crohn’s fistulas. Lancet. 2016.
Hare JM, et al. POSEIDON randomized trial. JAMA. 2012.
Steinberg GK, et al. MSC therapy for stroke. Stroke. 2016.
Wang D, et al. MSC therapy in autoimmune disease. Stem Cells Int. 2014.
Golpanian S, et al. Allogeneic human mesenchymal stem cell infusions for aging frailty. J Gerontol A Biol Sci Med Sci. 2017.
Sipp D. Japan’s conditional approval system for regenerative medicine. Nature. 2015.
Longeveron Inc. Company-reported Phase 2a clinical data presentation. 2024.
Perin EC, et al. Randomized Trial of Targeted Transendocardial Mesenchymal Precursor Cell Therapy in Patients With Heart Failure (DREAM-HF). J Am Coll Cardiol. 2023.
Xing D, et al. Efficacy and safety of mesenchymal stem cell therapy for knee osteoarthritis: a systematic review and meta-analysis. J Orthop Surg Res. 2024.
Perkins KM, et al. Infections After Receipt of Bacterially Contaminated Umbilical Cord Blood–Derived Stem Cell Products — United States, 2018. MMWR Morb Mortal Wkly Rep. 2018.
Medical Disclaimer: This blog is for educational purposes only and is not a substitute for medical advice. Always consult a qualified healthcare professional before starting or modifying any treatment.